EpiPatch™
“Flexibly fitting all shapes and sizes for seamless tympanic membrane repair.”


The tympanic membrane perforations repair system offers a versatile solution adaptable to different shapes and sizes of perforations. Its simplified procedure allows for in-office application under local anesthesia, eliminating the need for operating rooms and general anesthesia.
Devlopement and Compliance
BioMed’s EpiPatch, is developed from human amniotic tissue, selected for its natural elasticity and protective capabilities. This tissue, obtained following cesarean deliveries, undergoes decellularization to preserve its extracellular matrix (ECM).
These products comply with FDA guidelines, notably 21 CFR Part 1271, emphasizing their role as protective barriers that maintain elasticity, mirroring the amniotic tissue’s natural function. The decellularization process employs supercritical CO2 technology, which maintains the amniotic membrane’s structure and function, thus optimizing its therapeutic efficacy.
EpiPatch’s thinness, less than 0.1 mm, allows it to serve as a bioactive wound dressing that adheres upon contact, simplifying postoperative care, and is valued for its ease of use and storage.
EpiPatch Used With The MyringoPatch
EpiPatch In-Action

The bony surface was in a slightly wet state from blood or effusion.
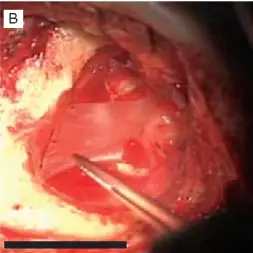
TA piece of the hyperdry AM was cut to the required size.

Hyperdry AM was slightly pressed to stick firmly to the bony surface (arrows).
Check Out Our Resource Center for All Certifications, FDA Regulations and Clinical Evidence
Download the BioMed app to share documents on-the-go!
Ordering Information
Code | Description | Diameter | Thickness |
BE2108FD | EpiPatch* Disc – S, Freeze Dried | 8 MM | 0.1 MM |
BE2110FD | EpiPatch* – M, Freeze Dried | 10 x 10 MM | 0.1 MM |
BE2010FD | EpiPatch* – Disc M, Freeze Dried | 10 MM | 0.1 MM |
BE2012FD | EpiPatch* – Disc L, Freeze Dried | 12 MM | 0.1 MM |
BE2120FD | EpiPatch* – L, Freeze Dried | 20 x 20 MM | 0.1 MM |
BE2016FD | EpiPatch* Disc – XL, Freeze Dried | 16 MM | 0.1 MM |
BE2060FD | EpiPatch* – XL, Freeze Dried | 20 x 30 MM | 0.1 MM |
* Medtronic EpiDisc and EpiFilm Replacement | |||