MyringoPatch™
“The Safe Alternative To Autologous Fascia Temporalis”
- Suturable Design: Its unique composition allows for direct suturing, enabling a precise placement and secure fixation during the healing period.
- Proven Clinical Success: MyringoPatch has demonstrated excellent clinical results, underscoring its reliability and effectiveness in patient care.
- Minimized Risk: By avoiding a secondary surgery site, MyringoPatch significantly reduces the chances of infection and scarring, promoting a smoother recovery process.
- Enhanced Mechanical Strength: MyringoPatch is crafted for durability, offering superior resistance to facilitate effective healing.


MyringoPatch is engineered to eliminate the need for a second surgery site, ensuring optimal surgical outcomes without compromise. With these advantages, MyringoPatch represents a significant advancement in the treatment of tympanic membrane perforations, offering patients a safer and more reliable healing journey.
MyringoPatch Used With The EpiPatch
Case Studies
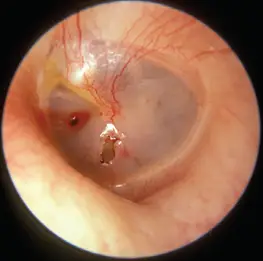
Tympanic membrane perforations vary significantly in shape and size, stemming from a multitude of causes. While complex cases often necessitate an underlay procedure conducted in the operating room under general anesthesia, not all situations require such extensive measures. Successful interventions may still result in residual or recurrent perforations, and for these revision cases—as well as some primary perforations of limited size—an in-office procedure with a transcranial overlay under local anesthesia is adequate. For these instances, MyringoPatch presents a biological solution, acting as a protective bandage to cover and safeguard the surgical area, streamlining the healing process without the need for operating room facilities.
Harvinder S, Hassan S, Sidek DS, Hamzah M, Samsudin AR, Philip R.
Underlay myringoplasty: comparison of Human Amniotic membrane to temporalis fascia graft.
Med J Malaysia. 2005 Dec; 60(5):585-9 PMID: 16515109
https://pubmed.ncbi.nlm.nih.gov/16515109/
Yung, M., Vivekanandan, S., Smith, P.
Randomized study comparing fascia and cartilage grafts in myringoplasty.
Ann. Otol. Rhinol. Laryngol, 2011; 120:535Y41
https://pubmed.ncbi.nlm.nih.gov/21922978/
Jalali. M.M., e.a.
Comparison of cartilage with temporalis fascia tympanoplasty: A meta-analysis of comparative studies
Laryngoscope, 2017; 127:2139-2148
https://pubmed.ncbi.nlm.nih.gov/27933630/
Healing Progression – Before and After (10 Weeks)

Check Out Our Resource Center for All Certifications, FDA Regulations and Clinical Evidence
Download the BioMed app to share documents on-the-go!
Ordering Information
Code | Description | Diameter | Thickness |
BE1010FD | MyringoPatch – M, Freeze Dried | 10 MM | 0.2 MM |
BE1016FD | MyringoPatch – L, Freeze Dried | 16 MM | 0.2 MM |
BE1030FD | MyringoPatch – XL, Freeze Dried | 25 x 30 MM | 0.2 MM |